SOPHiA DDM™ for Hereditary Cancers
Confidently assess genetic variants predisposing to cancer
Hereditary causes account for ∼10% of cancer cases, and an estimated 20% of cancer patients have a family history of cancer.1
Identification of an individual with a suspected hereditable cancer can lead to additional examinations and help formulate the most appropriate prevention strategies. Evaluating the predisposition to develop inherited cancer depends on the ability to characterize genes and alterations associated with increased cancer risk accurately. Next-generation sequencing (NGS) is transforming the way genomic evaluation of hereditary cancers is performed and integrated into the daily workflow of clinical laboratories.
Approximately 10-15% of breast cancer cases are associated with hereditary syndromes that carry a deleterious mutation in BRCA1/2 in most of the cases2
Mutations in the high-penetrance (i.e., TP53, PTEN, CDH1, MSH1, MLH1, MSH6, PMS2, PALB2, and STK11) and moderate-penetrance genes (i.e., CHEK2, ATM, and BRIP1) also correlate with high lifetime risks of breast cancers and other malignancies3.
Completing genetic testing is becoming essential for the complex care of patients with suspected hereditary breast and ovarian cancers (HBOC) and their at-risk family members. Genetic testing is increasingly provided through NGS multigene panels. However, the complexity of NGS data analysis and interpretation of variants are real-time challenges for its routine use4.
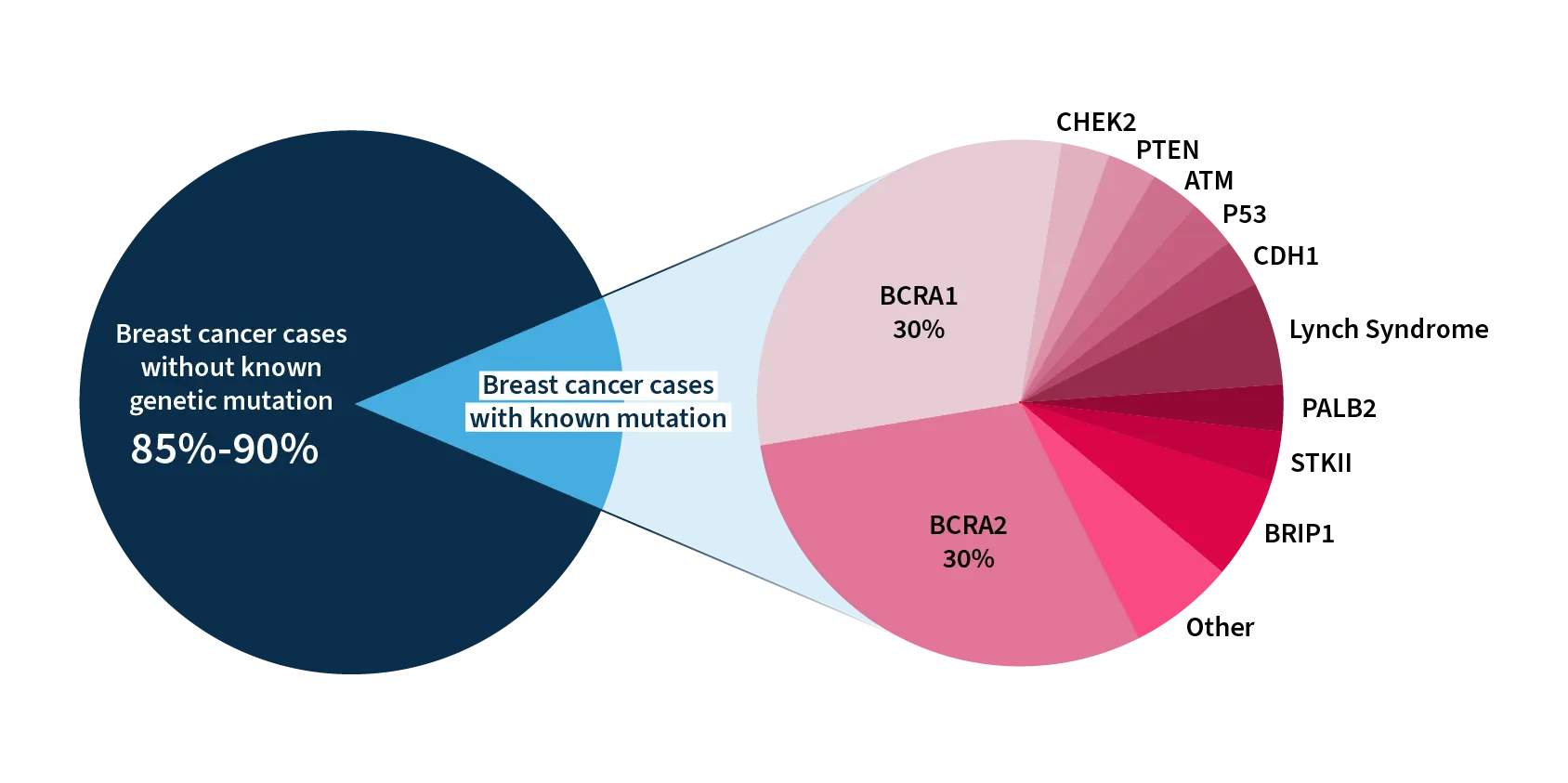
Breast cancer patients with genetic mutations.
Image from the paper: Comparison of Patient Susceptibility Genes Across Breast Cancer: Implications for Prognosis and Therapeutic Outcomes. Pharmacogenomics Pers Med. 2020;13:227-238
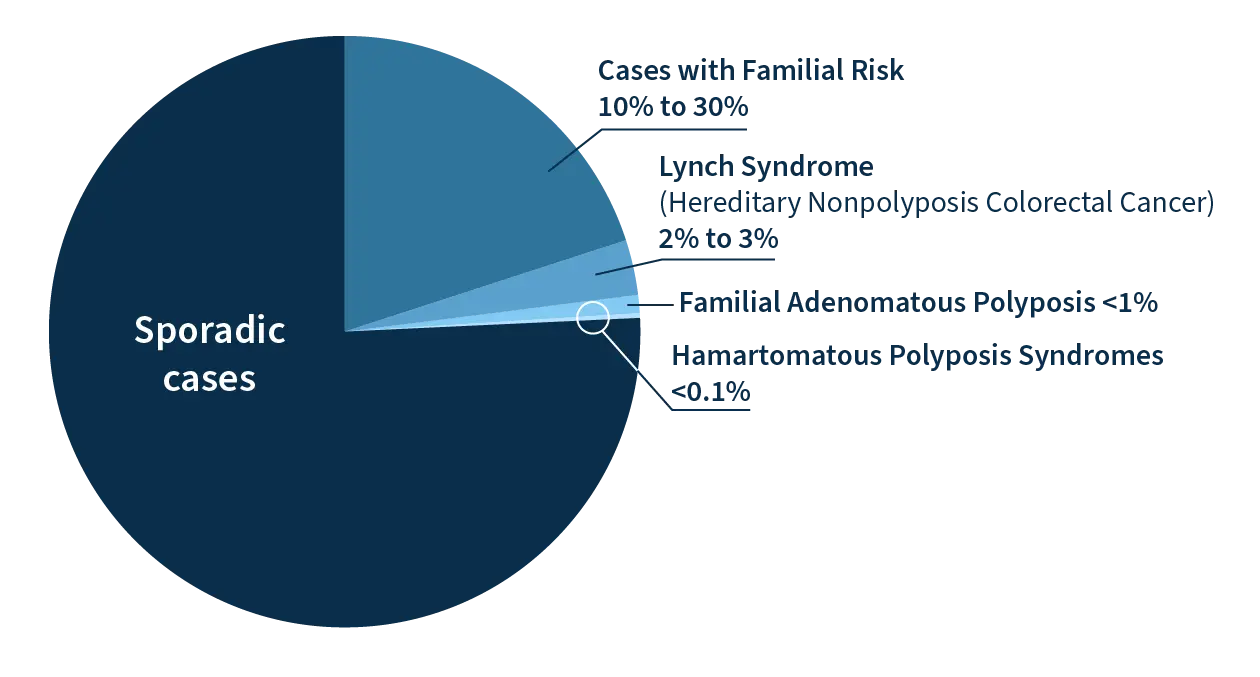
Fractions of colon cancer cases that arise in various family risk settings. Adapted from: Gastroenterology, Vol. 119, No. 3, Randall W. Burt, Colon Cancer Screening, Pages 837-853, Copyright (2000), with permission from Elsevier.
Recent data shows that colorectal cancer (CRC) is one of the most important causes of death worldwide.
Between 2% to 5% of all colorectal cancers (CRC) arise in the setting of well-defined inherited syndromes (e.g., Lynch syndrome, familial adenomatous polyposis, MUTYH-associated polyposis, and certain hamartomatous polyposis conditions).5 Each condition is associated with a high risk of developing colon cancer. Proper identification of predisposing genes allows for accurate risk assessment and more precise screening approaches.
SOPHiA DDM™ for Hereditary Cancers gives experts confidence in their results and overcomes challenges associated with analyzing the genes linked to the major hereditary cancer syndromes (i.e., breast and ovarian cancer, Lynch and intestinal polyposis syndromes).
Find the answers you need from your data.
In one unique experiment, the SOPHiA DDM™ Platform for hereditary cancers offers you:
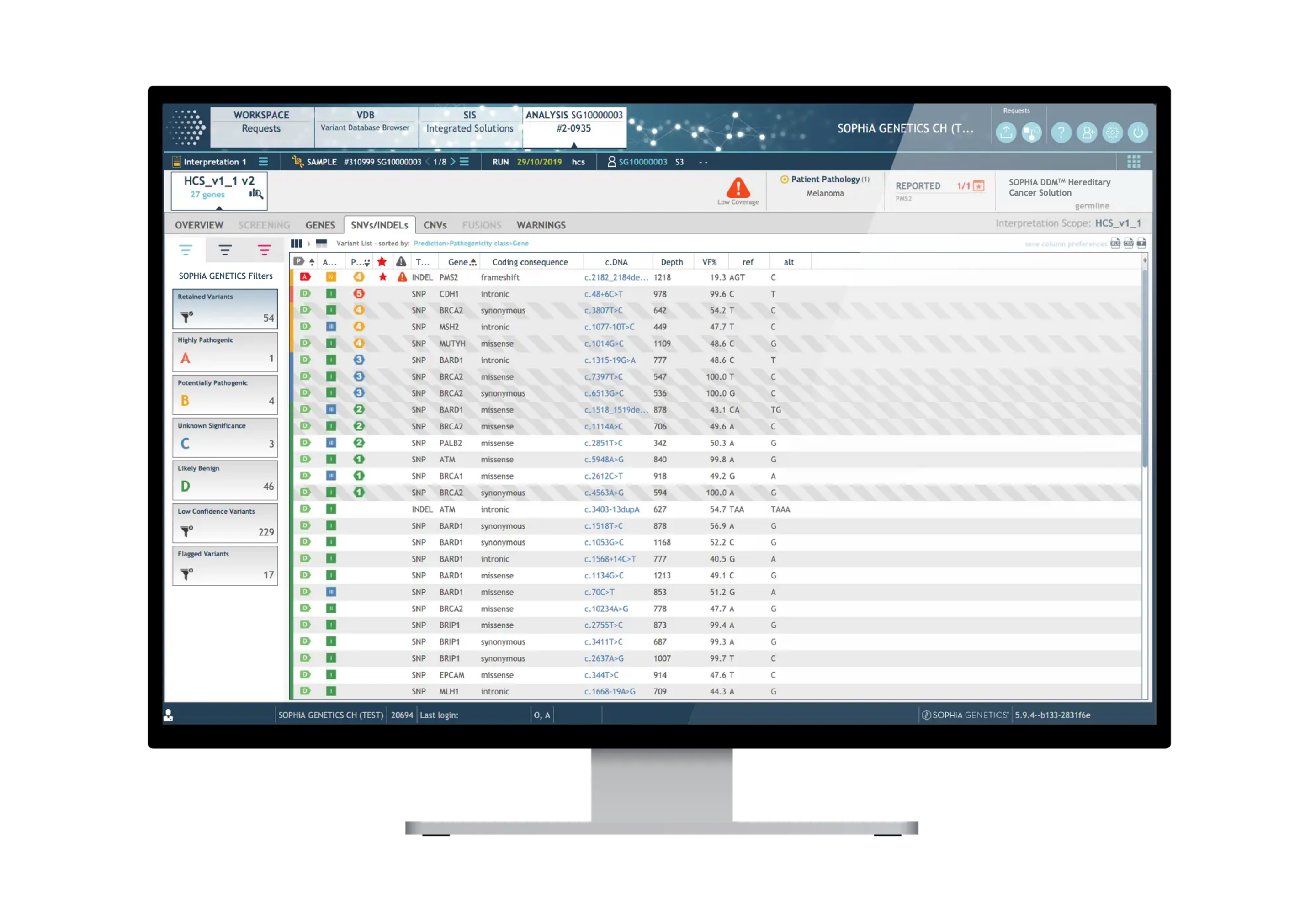
- Accurate detection of single-nucleotide variants (SNVs), small insertion or deletion events (Indels), and copy number variations (CNVs)
- Precise identification of Alu insertions, particularly abundant in BRCA16
- Confident discrimination between variants in PMS2CL from the causative ones in PMS2
- Reliable assessment of the Boland inversion in the MSH2 gene
The SOPHiA DDM™ Platform also provides several tools that help you categorize variants including population, disease, and, sequence databases, ACMG scoring and an algorithm-driven variant ranking (A, B, C, D). In addition, through SOPHiA DDM™ you have access to one of the largest networks of connected healthcare institutions worldwide to safely share knowledge and expertise with your peers, facilitating decision-making.
Relevant resources
Discover the solutions of SOPHiA DDM™ for Hereditary Cancers
SOPHiA GENETICS™ Hereditary Cancer Applications
SOPHiA DDM™ Dx Hereditary Cancer Solution

1 Al Harthi, F. S., et al. (2020) ‘Familial/inherited cancer syndrome: a focus on the highly consanguineous Arab population’, npj Genomic Medicine, 5, 3
2 Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA. 2017;317(23):2402–2416. doi:10.1001/jama.2017.7112
3 Peleg Hasson S, Menes T, Sonnenblick A. Comparison of Patient Susceptibility Genes Across Breast Cancer: Implications for Prognosis and Therapeutic Outcomes. Pharmacogenomics Pers Med. 2020;13:227-238 https://doi.org/10.2147/PGPM.S233485
4 Federici G, Soddu S. Variants of uncertain significance in the era of high-throughput genome sequencing: a lesson from breast and ovary cancers. J Exp Clin Cancer Res. 2020;39(1):46. doi:10.1186/s13046-020-01554-6
5 Kory W. Jasperson, Thérèse M. Tuohy, Deborah W. Neklason, Randall W. Burt, Hereditary and Familial Colon Cancer, Gastroenterology, Volume 138, Issue 6, 2010, Pages 2044-2058, ISSN 0016-5085, https://doi.org/10.1053/j.gastro.2010.01.054
6 Sharifah NA, Nurismah MI, Lee HC et al: Identification of novel large genomic rearrangements at the BRCA1 locus in Malaysian women with breast cancer. Cancer Epidemiol 2010; 34: 442–447
2 Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA. 2017;317(23):2402–2416. doi:10.1001/jama.2017.7112
3 Peleg Hasson S, Menes T, Sonnenblick A. Comparison of Patient Susceptibility Genes Across Breast Cancer: Implications for Prognosis and Therapeutic Outcomes. Pharmacogenomics Pers Med. 2020;13:227-238 https://doi.org/10.2147/PGPM.S233485
4 Federici G, Soddu S. Variants of uncertain significance in the era of high-throughput genome sequencing: a lesson from breast and ovary cancers. J Exp Clin Cancer Res. 2020;39(1):46. doi:10.1186/s13046-020-01554-6
5 Kory W. Jasperson, Thérèse M. Tuohy, Deborah W. Neklason, Randall W. Burt, Hereditary and Familial Colon Cancer, Gastroenterology, Volume 138, Issue 6, 2010, Pages 2044-2058, ISSN 0016-5085, https://doi.org/10.1053/j.gastro.2010.01.054
6 Sharifah NA, Nurismah MI, Lee HC et al: Identification of novel large genomic rearrangements at the BRCA1 locus in Malaysian women with breast cancer. Cancer Epidemiol 2010; 34: 442–447
Want to know more?
Get in touch with us.
Our client services team is on hand to help.